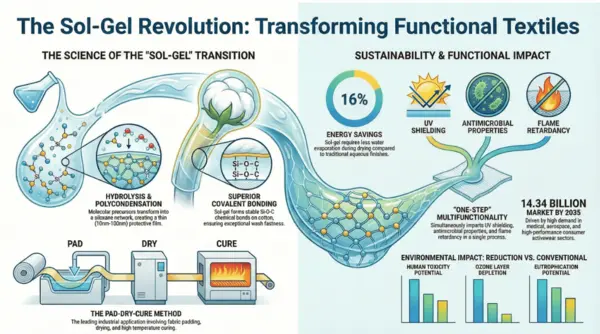
Sol-gel technology converts liquid precursors into structured inorganic coatings directly on textile fibers enabling flame retardancy, UV protection, antimicrobial activity, and hydrophobicity in a single processing step.
The process relies on hydrolysis and polycondensation of metal alkoxides primarily silicon-based — forming a nanoscale siloxane network that chemically bonds to fiber surfaces.
Compared to conventional wet finishing, sol-gel reduces energy consumption by 16% and avoids hazardous chemicals like formaldehyde and halogenated flame retardants.
The technology is gaining commercial traction in medical, defense, and performance apparel sectors with global sol-gel market value projected to reach $14.34 billion by 2035.
Introduction: Why Sol-Gel Technology Matters in Modern Textile Engineering
The pressure on textile engineers today is unlike anything the industry has faced before. Mills are expected to deliver fabrics that are multifunctional, durable, compliant with tightening chemical regulations, and produced with a reduced environmental footprint all at competitive cost.
Traditional wet finishing processes struggle to meet these demands simultaneously. They rely on large volumes of water, energy-intensive drying stages, and chemical cross-linkers like formaldehyde that are hazardous to workers and restricted by global standards.
Sol-gel technology offers a fundamentally different approach. Instead of applying functional agents through conventional padding chemistry, it builds a structured inorganic or hybrid organic-inorganic network directly on the fiber surface at the nanoscale.
First observed in the ceramics and glass sector as far back as 1846, sol-gel remained outside textile engineering for over a century. The last two decades have changed that entirely. Today, it is recognized as one of the most promising platforms for high-performance textile finishing.
This guide breaks down the core chemistry, industrial application methods, functional capabilities, substrate interactions, sustainability profile, and commercial landscape of sol-gel technology written specifically for engineers and technicians who need technical depth, not surface-level summaries.
The Core Chemistry of Sol-Gel Technology
Understanding sol-gel starts with its two fundamental chemical reactions: hydrolysis and polycondensation. These reactions transform metal alkoxide precursors most commonly silicon-based compounds like tetraethyl orthosilicate (TEOS) or tetramethyl orthosilicate (TMOS) into a three-dimensional inorganic oxide network.
Step 1: Hydrolysis
In the presence of water and a catalyst, alkoxy groups (–OR) attached to the silicon atom are progressively replaced by hydroxyl groups (–OH), generating silanol moieties. The general reaction takes the form:
Si(OR)₄ + nH₂O → Si(OR)₄₋ₙ(OH)ₙ + nROH
Acid catalysts are preferred for textile applications because they drive the formation of linear, weakly branched structures ideal for producing thin, uniform films on fiber surfaces. Base catalysts, by contrast, generate dense, highly branched clusters better suited for ceramic or optical applications.
Step 2: Polycondensation
As silanol groups accumulate, they react with each other or with remaining alkoxy groups to form siloxane bridges (Si–O–Si). Two parallel condensation pathways operate simultaneously:
- Water condensation: ≡Si–OH + ≡Si–OH → ≡Si–O–Si≡ + H₂O
- Alcohol condensation: ≡Si–OH + ≡Si–OR → ≡Si–O–Si≡ + ROH
The continuous growth of these siloxane linkages produces colloidal particles the sol which eventually crosslink into a three-dimensional gel network enclosing the liquid phase.
Critical Process Variables
Controlling the kinetics and final morphology of the sol-gel coating requires precise management of several process parameters:
- Water-to-precursor molar ratio: Higher ratios accelerate hydrolysis but risk excessive shrinkage and cracking during drying and curing.
- Catalyst choice: Acid catalysts produce polymer-like linear chains; base catalysts produce dense branched clusters.
- Temperature: Drying typically occurs at 80–100°C; curing completes network stabilization at up to 150°C.
- Precursor composition: Hybrid organic-inorganic precursors enable tailored surface chemistry for specific functional outcomes.
The resulting coating typically between 10 nm and 100 nm thick forms a dense nanocomposite polymer film on individual fiber surfaces without significantly altering fabric hand or breathability.
Industrial Application Techniques for Sol-Gel on Textiles
Choosing the right deposition method is as critical as formulating the sol itself. The method determines coating uniformity, penetration depth, layer thickness, and production throughput.
Pad-Dry-Cure: The Industry Standard
The pad-dry-cure process dominates large-scale textile finishing with sol-gel. The workflow is straightforward:
- Fabric passes through a bath containing the sol formulation.
- Rollers squeeze the fabric to ensure uniform sol uptake across the full width.
- The fabric enters a dryer (80–100°C) to remove solvent and water.
- Final curing (up to 150°C) consolidates the siloxane network and drives covalent bonding to the fiber surface.
On cellulosic fibers like cotton, the curing stage drives the formation of covalent Si–O–C bonds between silanol groups and fiber hydroxyl groups delivering excellent wash fastness. On synthetic fibers like polyester and polyamide, adhesion is achieved mainly through hydrogen bonds and physical encapsulation within the fiber interstices.
Spray-Coating and Rolling
For technical textiles with complex three-dimensional geometries or asymmetric coating requirements, spray-coating provides greater flexibility. This technique allows for controlled nanostructure formation and is particularly effective for applying functionalized hydrogels or conductive inks to substrates destined for wearable electronics applications.
Rolling and dip-coating offer different penetration profiles and layer thicknesses. Dip-coating is favored for multi-layer assemblies such as antireflective or self-cleaning surfaces where sequential layer deposition is required.
Spin-Coating for Specialty Applications
Spin-coating is not suited to continuous production but delivers extremely high uniformity for specialized substrates. It is primarily used in the development of optical sensors and smart display textiles where sub-micron uniformity is a hard requirement.
Scalability Challenges
Transitioning from laboratory scale to mass production introduces several engineering hurdles that technicians must account for:
- Slow gelation and drying: Can bottleneck high-speed production lines. Process optimization is required to balance line speed with adequate network formation.
- Precursor stability: Commercial nanosols require purification typically through anion exchange resins to ensure batch-to-batch consistency at industrial volumes.
- Capital investment: Certain advanced sol-gel variations require specialized reactors and vacuum systems, presenting barriers for small to medium enterprises.
Multifunctional Properties Achievable with Sol-Gel Textile Finishing
The defining commercial advantage of sol-gel is its capacity to deliver multiple protective functions from a single finishing step by incorporating different nanoparticles, functional precursors, or organic modifiers directly into the sol formulation.
Flame Retardancy
Silica-based sol-gel networks act as a physical thermal barrier on the fiber surface. During combustion, this layer limits heat flux and restricts oxygen diffusion to the substrate delaying ignition and slowing flame spread.
More advanced formulations incorporate phosphorus- and nitrogen-doped silanes, which promote stable char formation on cotton fibers during burning. This synergistic effect reduces the peak heat release rate (pHRR) and prevents fabric dripping critical performance criteria for firefighter uniforms, industrial protective workwear, and military applications.
Crucially, these effects are achieved without halogenated compounds which are restricted under multiple international environmental regulations.
Hydrophobicity and Oleophobicity
Water and oil repellency is engineered through alkyl-modified silanes such as hexadecyltrimethoxysilane or octyltriethoxysilane (OTES) or through fluorochemical agents incorporated into the sol formulation.
These treatments lower the surface energy of the fiber and create nanoscale roughness mimicking the lotus effect. Hybrid formulations for example, OTES combined with melamine-formaldehyde have achieved water contact angles of up to 150° on polyester, rendering it superhydrophobic while maintaining breathability.
Antimicrobial and Antiviral Activity
Potent antimicrobial functionality is achieved by embedding metal oxide nanoparticles particularly zinc oxide (ZnO) and titanium dioxide (TiO₂) or colloidal silver (Ag) into the sol-gel matrix.
These systems are especially valuable in healthcare textiles hospital gowns, surgical drapes, laboratory coats where prevention of microbial colonization is a primary performance requirement. TiO₂ provides additional photocatalytic antimicrobial activity beyond direct contact mechanisms.
UV Shielding and Photocatalytic Self-Cleaning
Sol-gel coatings embedding TiO₂ or ZnO absorb and scatter UV radiation, delivering a high Ultraviolet Protection Factor (UPF). This is particularly relevant for outdoor workwear and activewear where extended UV exposure is a concern.
TiO₂ also enables photocatalytic self-cleaning. When exposed to light with energy exceeding its band gap (approximately 3.2 eV), TiO₂ generates reactive oxygen species primarily hydroxyl radicals that degrade organic contaminants, bacteria, and odor molecules into water and carbon dioxide. This mechanism is reversible and reduces laundering frequency over the garment lifecycle.
Substrate Interaction and Durability: Cotton vs. Synthetic Fibers
The longevity of any sol-gel finish depends entirely on the quality of chemical adhesion between the coating and the substrate. Fiber chemistry is the determining factor.
Cotton and Cellulosic Fibers
Cotton offers the most favorable surface chemistry for sol-gel adhesion. The abundant hydroxyl groups on the cellulose backbone react with silanol moieties during curing to form stable covalent Si–O–C linkages. This results in strong wash fastness treatments on cotton can maintain their functional properties across many laundry cycles under optimized processing conditions.
Polyester and Polyamide
Polyester (PES) presents the most challenging substrate for sol-gel adhesion. With a very low moisture regain of approximately 0.4% and few reactive surface sites, bonding relies predominantly on hydrogen bonds and physical encapsulation within the fiber structure.
Polyamide (PA/Nylon) is more accommodating with a moisture regain of approximately 4.5% and amide bonds that offer better hydrogen bonding potential. However, both synthetic fibers risk coating delamination over extended use unless surface activation techniques particularly plasma treatment are employed prior to sol-gel application.
Despite adhesion limitations on synthetics, sol-gel coatings have been shown to improve tensile strength and anti-crease properties on both polyester and polyamide. Abrasion resistance may decrease slightly in certain silica-treated cotton fabrics.
Technical Textiles: Aramid Performance Data
For aramid fabrics used in ballistic protection, sol-gel alumina (Al₂O₃) coatings have demonstrated notable improvements: frictional energy dissipation and impact resistance increased by up to 35% while bending stiffness was reduced by only 5%. This balance of performance enhancement with minimal hand change is a key engineering advantage in technical textile applications.
Sol-Gel in Smart and Intelligent Textile Systems
Beyond static functional finishes, sol-gel chemistry serves as an enabling platform for the next generation of responsive and interactive textiles.
Thermochromic and Temperature-Regulating Materials
Sol-gel matrices are used to encapsulate leuco dye-based thermochromic pigments, providing stable color-change behavior in response to temperature variations. These systems have practical applications in medical monitoring detecting fever through visual color shift without electronic sensing components.
Phase-Change Materials (PCMs) such as tetradecyl alcohol can be incorporated into the sol-gel matrix to create thermoregulating fabrics. In wool substrates, PCM-loaded sol-gel composites have demonstrated latent heat storage capacity of up to 89.2 J/g. The silica network physically binds the PCM, preventing leakage during phase transition while maintaining fabric flexibility.
Wearable Electronics and Sensor Integration
Sol-gel acts as a carrier and encapsulant for conductive inks, flexible sensors, and electronic components integrated into fabric structures. Key developments include:
- Conductive yarns: Cotton yarns treated with carbon nanotube solutions or polymer-based conductive inks enable power transport to embedded LED devices or biometric sensors.
- Biometric apparel: Compression garments with integrated sensors capable of measuring heart rate, muscle exertion, and postural data in real time.
- Energy harvesting: Triboelectric and thermoelectric nanogenerators embedded in sol-gel-treated fabrics generate power from body movement or heat — reducing dependence on external batteries in wearable technology.
Sustainability Profile and Life Cycle Analysis
For textile engineers operating under corporate sustainability mandates, the environmental credentials of sol-gel technology are among its strongest arguments.
Energy and Resource Efficiency
Life Cycle Analysis comparing sol-gel processing to conventional pad-dry-cure finishing has identified a 16% reduction in energy consumption during the drying phase. The primary driver is reduced water content in the sol relative to conventional aqueous finishing baths — meaning less energy is required to achieve dryness.
Where organic co-solvents like isopropanol are used, Abiotic Depletion and Global Warming Potential scores increase. This has accelerated research into water-based sol-gel dispersions that minimize VOC emissions while retaining functional performance.
Chemical Safety Advantages
Sol-gel chemistry is inherently compatible with formaldehyde-free finishing. Formaldehyde a Category 1 carcinogen under IARC classification is widely used as a cross-linking agent in conventional easy-care and wrinkle-resistant finishes. Sol-gel eliminates this chemistry entirely.
The high durability and wash fastness of sol-gel coatings mean that fewer functional nanoparticles are released into wastewater during consumer laundering. This directly reduces the risk of nanoparticle bioaccumulation in aquatic ecosystems an increasingly significant concern under EU REACH and similar frameworks.
Market Landscape and Industry Players
The global sol-gel product market was valued at $9.83 billion in 2024 and is projected to grow to $14.34 billion by 2035. While the electronics and automotive sectors drive the largest share of this growth, textile applications are expanding particularly in healthcare, defense, and performance apparel.
Key Industry Stakeholders
Understanding the commercial ecosystem helps engineers navigate sourcing, specification, and partnership decisions:
- Multinational Suppliers: 3M, Solvay, BASF, and Huntsman Corporation supply advanced silane precursors and metal oxide nanoparticles for sol-gel formulations.
- Specialized Coating Firms: Socomore’s SOCOGEL product range delivers adhesion promoters and protective coatings qualified by major aerospace manufacturers including Boeing and Airbus.
- Textile Brands: Schoeller Textiles (NanoSphere), Crypton, Momentum, and Carnegie Fabrics are established adopters of nanotechnology-based sol-gel finishes.
- Research Institutions: Saarland University (Germany), STRIDE (Malaysia), and Universiti Malaysia Pahang are active in developing water-based sol-gel methods for military and medical applications.
Highest-Growth Sectors
Within textiles, three segments are driving the strongest adoption:
- Medical and Healthcare: Antimicrobial surgical textiles and regenerative tissue scaffolds are growing at a reported CAGR of 11.2% within the medical sol-gel segment.
- Aerospace and Defense: Heat-resistant uniforms and sol-gel-reinforced ballistic fabrics for armored applications.
- Consumer Activewear: Major brands are integrating conductive fibers and multifunctional finishes into mainstream performance garments.
Key Takeaways
- Chemistry precision is critical: Hydrolysis rate, catalyst type, and water-to-precursor ratio all directly determine coating morphology, adhesion strength, and functional performance.
- Substrate chemistry defines durability: Cotton provides covalent Si–O–C bonding for superior wash fastness; synthetics require surface activation (e.g., plasma treatment) for comparable adhesion.
- Multifunctionality in one step: Flame retardancy, hydrophobicity, antimicrobial activity, UV shielding, and self-cleaning can be achieved simultaneously by tailoring the sol formulation.
- Sustainability advantages are measurable: 16% drying energy reduction, formaldehyde-free chemistry, and reduced nanoparticle laundering release versus conventional finishes.
- Scalability hurdles remain: Slow gelation, precursor stability management, and capital costs for advanced variants are engineering challenges requiring active management.
- Smart textile integration is the frontier: Sol-gel serves as the enabling chemistry for thermochromic coatings, PCM fabrics, wearable sensors, and energy harvesting systems.
FAQ: Sol-Gel Technology in Textile Finishing
Q: What is sol-gel technology and how is it used in textiles?
Sol-gel technology converts liquid molecular precursors primarily silicon alkoxides into structured inorganic coatings through hydrolysis and polycondensation reactions. In textiles, these coatings are applied to fiber surfaces using methods like pad-dry-cure to impart properties such as flame retardancy, UV protection, and antimicrobial activity.
Q: How does sol-gel technology compare to conventional textile finishing in terms of environmental impact?
Sol-gel offers measurable environmental advantages: it eliminates formaldehyde-based cross-linkers, achieves approximately 16% lower energy use in drying, and produces more durable coatings that release fewer nanoparticles during laundering. Water-based sol-gel formulations further reduce VOC emissions versus solvent-based alternatives.
Q: Why is sol-gel coating more effective on cotton than on polyester?
Cotton’s cellulose structure provides abundant hydroxyl groups that react with sol silanol moieties to form covalent Si–O–C bonds during curing delivering strong, durable adhesion. Polyester has very low moisture regain (~0.4%) and few reactive surface sites, limiting bonding to hydrogen interactions and physical encapsulation, which are less durable over laundering cycles.
Q: What are the key process variables that textile technicians must control in sol-gel finishing?
Critical variables include: water-to-precursor molar ratio (affects hydrolysis rate and shrinkage risk), catalyst type and concentration (controls network morphology), drying temperature (80–100°C) and curing temperature (up to 150°C), and precursor selection based on the target functional property.
Q: Can sol-gel coatings provide flame retardancy without halogenated chemicals?
Yes. Silica-based sol-gel networks provide flame retardancy through physical oxygen and heat barrier formation. Phosphorus- and nitrogen-doped silane formulations further promote stable char layer formation on cotton achieving performance equivalent to halogenated systems without the associated toxicity and regulatory restrictions.
Q: What is the current market size of the sol-gel industry and how significant is the textile segment?
The global sol-gel market was valued at $9.83 billion in 2024, with projected growth to $14.34 billion by 2035. The textile segment, while not the largest contributor, is experiencing stable growth driven particularly by the medical, aerospace/defense, and performance activewear sectors.
Conclusion
Sol-gel technology has moved decisively from laboratory science to industrial practice and for good reason. It addresses the three most pressing challenges in modern textile finishing: delivering high-performance functionality, achieving regulatory-compliant chemistry, and reducing environmental impact.
For textile engineers and technicians, the opportunity lies in understanding the chemistry precisely enough to control it. Precursor selection, catalysis, molar ratios, and curing conditions are not abstract variables they are the levers that determine whether a coating achieves durable multifunctionality or fails prematurely in the wash.
As water-based sol-gel routes mature and precursor costs decrease, expect adoption to accelerate across healthcare, defense, and consumer performance markets. The convergence with IoT and wearable technology signals that sol-gel will not merely be a finishing chemistry it will be the structural foundation of the next generation of intelligent textiles.
For more technical guides, process deep-dives, and industry insights, explore TextileInfoHub.com your professional resource for the global textile industry.